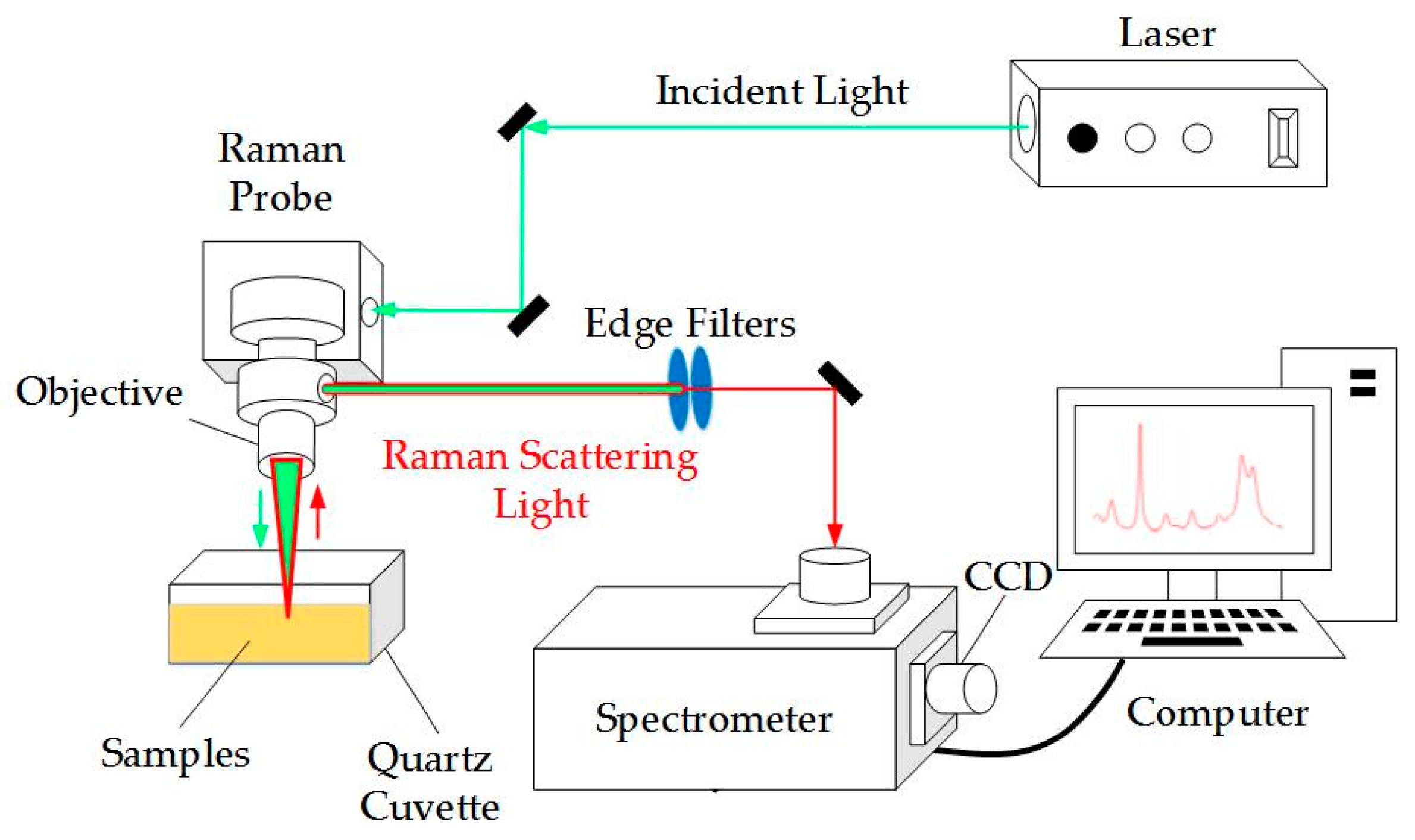
Raman Spectrometer Diagram at Karen Spaulding blog
Molecular Fingerprinting and the Technical Mechanics of the Raman Method
Imagine being able to tell the difference between a diamond and a piece of cubic zirconia without even touching them. Better yet, imagine identifying a dangerous chemical inside a sealed glass bottle without ever opening it. That is the specialized magic of the Raman Method. It is basically a secret handshake between a laser and a molecule. Honestly? It is one of the most underrated tools in the modern scientific arsenal, yet it is everywhere once you know what to look for.
I have spent over a decade staring at spectral lines, and let me tell you, they never lie. When you hit a substance with a high-intensity laser, most of the light just bounces off like a tennis ball hitting a wall. But a tiny, tiny fraction of that light does something weird. It exchanges energy with the molecules, changing color in the process. This specific phenomenon is the core of Raman spectroscopy, and it provides a unique chemical “fingerprint” that is impossible to forge.
Look—it is not just about fancy lasers and white lab coats. The Raman Method is about solving real-world puzzles. Whether we are talking about verifying the purity of a life-saving drug or figuring out if a Renaissance painting is a clever fake, this technique is the ultimate truth-teller. It is fast, it is non-destructive, and it is incredibly precise if you know how to read the data. Most people think science is all about messy test tubes, but this is clean, light-based detective work.
It is a big deal because it allows us to “see” molecular vibrations. Every molecule dances to its own rhythm. When we use the Raman Method, we are essentially listening to that dance. By measuring how the light shifts, we can identify exactly what atoms are bonded together and how they are moving. It sounds like science fiction, but it is actually just very clever physics that has been refined over nearly a century.
Foundational Principles Behind the Raman Method
To really get what the Raman Method is doing, we have to talk about scattering. When light hits a surface, most of it undergoes Rayleigh scattering, which is elastic. That means the light comes out with the same energy it went in with. Think of it like a mirror reflection; the color does not change. But Sir C.V. Raman discovered that about one in every ten million photons does something different. It loses or gains energy, a process we call inelastic scattering.
This energy shift is the “smoking gun” of molecular identity. When a photon interacts with a molecule, it can kick a molecular bond into a higher vibrational state. This takes energy away from the photon, shifting its color toward the red end of the spectrum. Conversely, if the molecule is already vibrating, it can give energy to the photon, shifting it toward the blue. We call these Stokes and Anti-Stokes shifts, and they are the bread and butter of the Raman Method.
Seriously, the precision here is wild. Because every molecule has a unique set of vibrational modes, the pattern of these shifts acts as a 100% unique identifier. You can distinguish between two chemicals that look identical to the naked eye, or even between different crystalline forms of the same substance. In my years in the lab, I have seen the Raman Method identify contaminants that were present in such small amounts they should have been invisible. It is like having microscopic X-ray vision, but for chemistry.

Raman Spectroscopy and Its Applications
The beauty of the Raman Method lies in its simplicity of setup compared to other techniques. You do not need to dissolve your sample in acid or turn it into a gas. You just point the laser and collect the light. However, the hardware involved is quite sophisticated. You need a stable laser source, a very high-quality filter to block out the “boring” Rayleigh light, and a sensitive detector to catch those rare, shifted photons. It is a game of filtering out the noise to find the signal.
Mechanisms of Inelastic Scattering
The actual interaction happens at the level of the electron cloud. When the laser hits the molecule, it induces a virtual state, essentially distorting the cloud of electrons. This distortion is what leads to the Raman scattering effect. If the molecule’s polarizability changes during a vibration, you get a signal. If it doesn’t, the Raman Method sees nothing. This is why it is so complementary to Infrared (IR) spectroscopy; they see different things.
I often tell my juniors that Raman spectroscopy is like feeling the texture of a molecule with light. The laser is the finger, and the shift is the sensation of the bumps and ridges. Because different bonds (like Carbon-Hydrogen versus Carbon-Oxygen) have different “textures,” they produce different shifts. This allows us to map out the entire structure of a complex organic compound in a matter of seconds. It’s incredibly efficient.
Interpreting the Raman Shift
The data we get back is usually a graph with “Raman Shift” on the x-axis and “Intensity” on the y-axis. These peaks correspond to specific molecular bonds. For instance, a big peak around 2900 cm-1 usually means you have got C-H bonds. A sharp spike at 1000 cm-1 might indicate a benzene ring. For an experienced specialist, reading these graphs is like reading a book. You start to see the story of the molecule unfolding right in front of you.
But here—and this is the kicker—you have to watch out for fluorescence. Some samples glow like a Christmas tree when you hit them with a laser, which can completely drown out the Raman Method signal. This is the bane of my existence. To get around it, we often change the laser wavelength. It’s a constant battle between getting a strong signal and avoiding the “noise” of a sample that just wants to glow. It keeps the job interesting, to say the least.
Practical Applications Across Modern Industry
One of the coolest things about the Raman Method is how it has escaped the basement of physics departments. Today, it is used on the factory floor. In the pharmaceutical industry, we use vibrational spectroscopy to ensure that the active ingredient in a pill is actually what it says on the box. We can even check if the powder is the right “polymorph,” which is a fancy way of saying the atoms are packed in the right shape. If the packing is wrong, the drug might not dissolve correctly in your body.
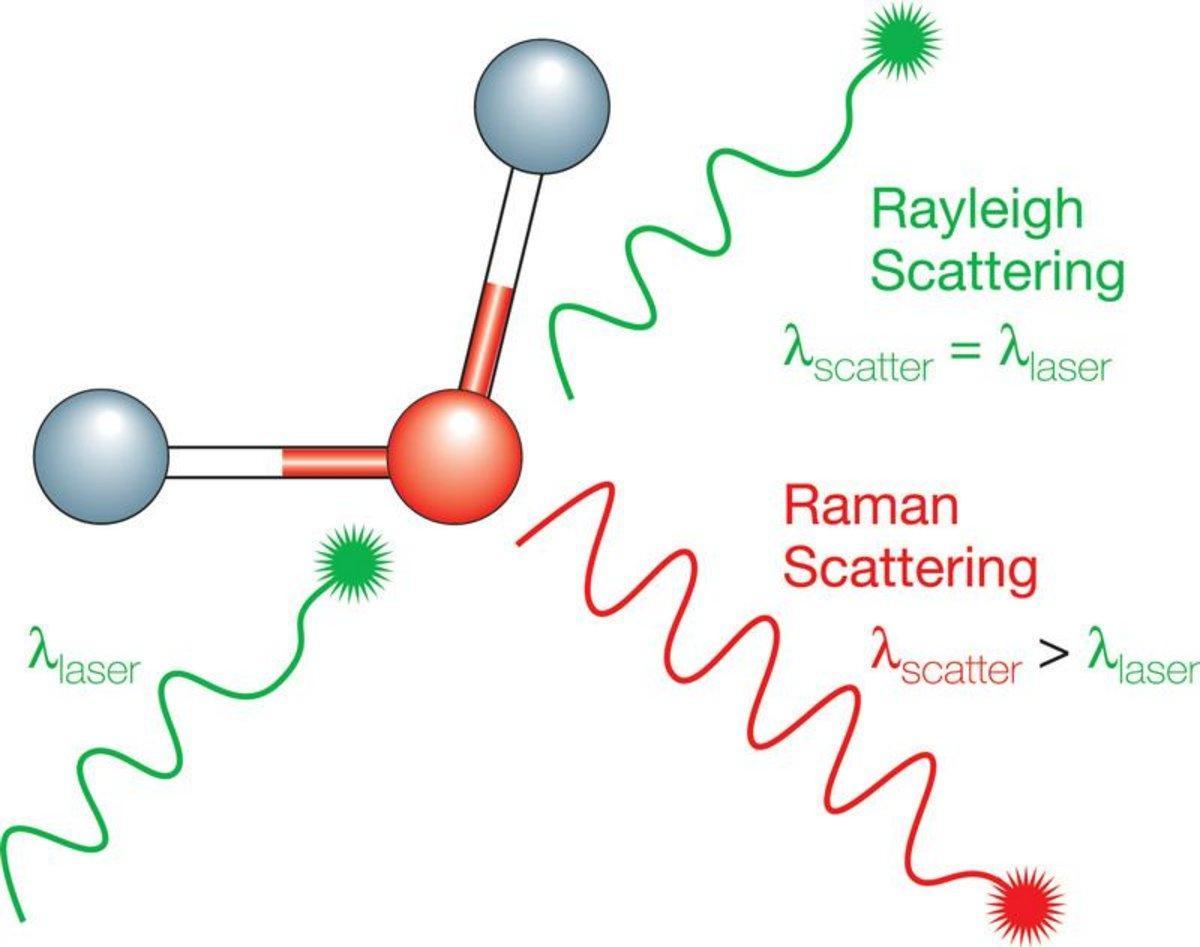
What are the Raman effect and Raman scattering?
Then there is the world of security and forensics. Have you ever seen those handheld devices that police use to test unknown powders? Many of those are portable Raman spectrometers. Because the Raman Method can “see” through clear plastic bags and glass vials, officers can identify a substance without ever risking exposure to it. It is literally a life-saver in the field. I have worked on calibration for these devices, and the accuracy is staggering for something that fits in your hand.
In the world of art and archaeology, the Raman Method is a game-changer because it is non-destructive. You can’t exactly take a chunk out of a 500-year-old fresco to test the pigment. But you can shine a low-power laser on it. We use it to identify the exact minerals used in ancient paints, which helps us date the work and plan conservation efforts. It is a bridge between the hard sciences and the humanities that I find absolutely fascinating.
Materials science also leans heavily on this. Carbon nanotubes, graphene, and other “wonder materials” are analyzed almost exclusively using the Raman Method. It tells us how many layers of graphene we have or if the nanotubes have defects. It is the gold standard for carbon-based materials. If you are working in tech or semi-conductors, you are likely using Raman scattering data every single day to verify your processes. It’s the silent workhorse of the silicon age.
Pharmaceutical Quality Control
-
- Active Ingredient Verification: Ensures the potency and identity of drugs during the manufacturing process.
- Contaminant Detection: Identifies foreign substances or impurities at very low concentrations.
- Polymorph Screening: Distinguishes between different crystal structures of the same chemical compound.
- Real-time Monitoring: Allows for continuous analysis during chemical synthesis to ensure reaction completion.

What Is Raman Spectroscopy at Michael Doxey blog
Art Conservation and Forensics
When dealing with a potential forgery, the Raman Method is the first line of defense. If a painting supposedly from the 17th century contains Titanium White—a pigment not invented until the 20th century—you know you have a fake. It provides objective, undeniable evidence. In my experience, art historians are often stunned by how much a simple laser can tell them about the history of a single brushstroke.
On the forensic side, it is used for everything from analyzing explosive residues to identifying ink on a forged check. Because it is so sensitive, we can get a signal from just a few grains of material. It is also great for analyzing body fluids at a crime scene without destroying the DNA evidence. It is a multi-purpose tool that has completely revolutionized how we process physical evidence. Honestly, it makes the stuff you see on TV look primitive.
The Practical Edge of the Raman Technique
Why do we choose the Raman Method over other options? The biggest reason is the lack of sample preparation. In most types of chemical analysis, you have to grind, dissolve, or stain your sample. That takes time and ruins the sample. With Raman spectroscopy, you can often just put the sample under the lens and go. This “point-and-shoot” capability is what makes it so valuable for high-throughput environments like industrial plants or airports.
Another major advantage is that water does not interfere with the signal. In Infrared spectroscopy, water is a nightmare because it absorbs IR light like a sponge, drowning out everything else. But the Raman Method is relatively blind to water. This makes it perfect for biological samples, like looking at live cells or analyzing chemicals in a liquid solution. It allows us to study life in its natural, wet state, which is a massive win for biochemistry.
Speed is also a massive factor. We are talking about results in seconds, not hours. In a modern lab, I can run a Raman analysis while I am still finishing my first cup of coffee. This rapid feedback loop allows scientists to iterate faster and catch mistakes before they become expensive problems. In a world where time is money, the Raman Method is an incredibly efficient investment for any research or production facility. It just works.
Finally, there is the versatility of the equipment. We have gone from massive, room-sized machines to handheld units and even microscopic versions. Micro-Raman allows us to focus the laser down to a single micrometer. This means we can analyze a single cell or a tiny inclusion in a gemstone. The sheer scale of what we can study—from huge industrial batches to microscopic particles—is what keeps the Raman Method at the top of the hierarchy of analytical techniques.
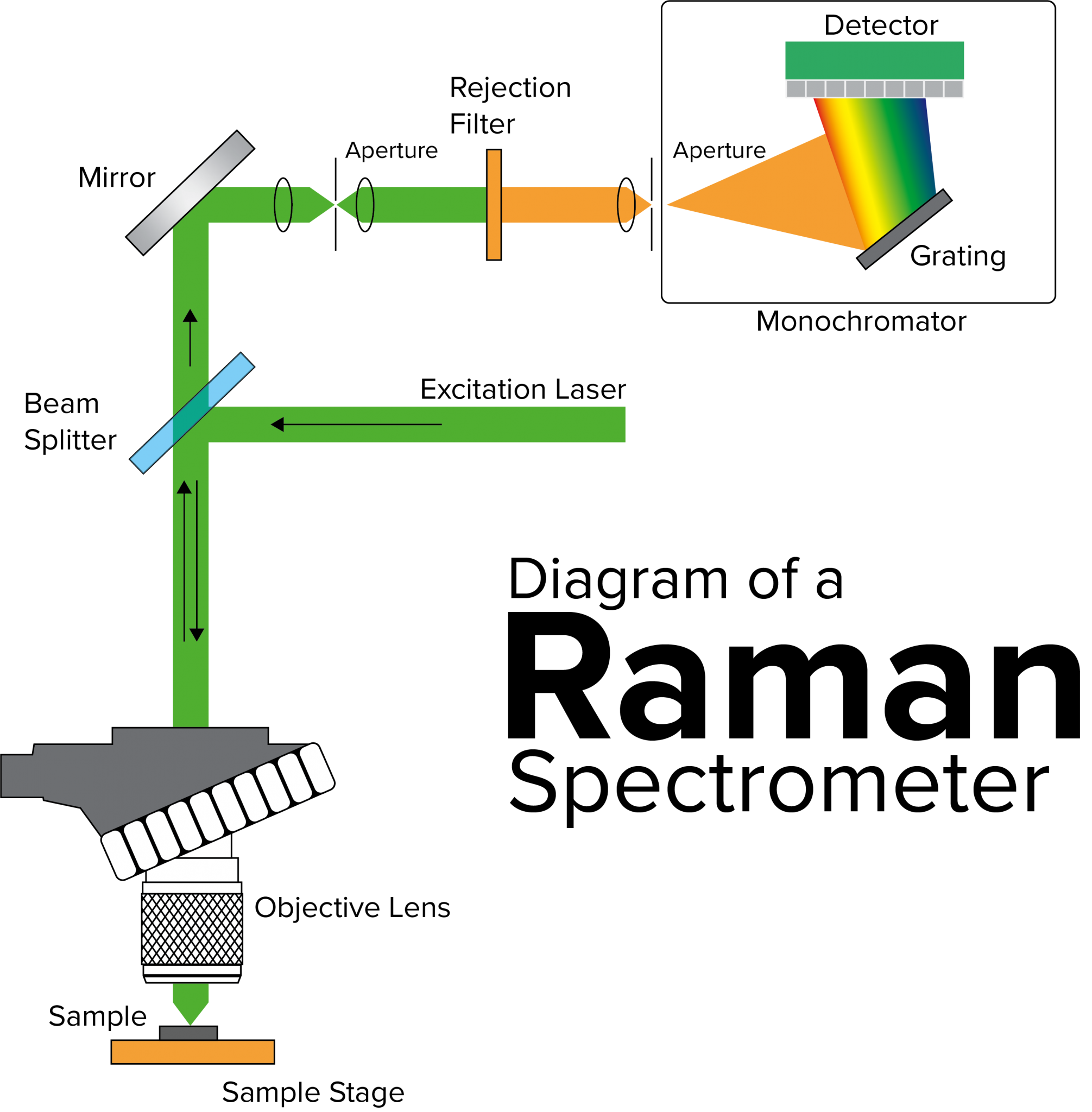
What is Raman Imaging? | JASCO
Non-Destructive Testing Benefits
- Sample Preservation: The material remains unchanged after analysis, allowing for further testing or sale.
- Analysis Through Packaging: Lasers can penetrate clear containers, maintaining a sterile environment.
- In Situ Measurements: Can be performed directly in the field or on the production line without moving the sample.
Speed and Preparation Efficiency
In the old days, you’d spend three hours preparing a sample and ten minutes analyzing it. The Raman Method flipped that script. Now, you spend zero minutes preparing and maybe thirty seconds analyzing. That shift in workflow has changed the culture of the laboratory. It allows us to be more curious and run more tests because the “cost” of a single test is so low. It encourages better science through higher volume.
It is also remarkably easy to automate. You can set up a robotic arm to feed samples into a Raman spectrometer and have it run 24/7. This is how modern manufacturing maintains such high standards of quality. By integrating the Raman Method directly into the conveyor system, we can catch a bad batch of material before it ever leaves the factory floor. It is proactive rather than reactive science, and that makes all the difference.

Basics of chemistry : From Light to Knowledge, Raman Spectroscopy in Action
Common Questions About The Raman Method
Is the Raman Method better than Infrared (IR) Spectroscopy?
It is not necessarily “better,” but it is different and often more convenient. While IR is great for identifying certain functional groups, the Raman Method excels at looking at the “backbone” of molecules and works much better with samples in water or inside glass containers. Most experts use them as complementary tools rather than choosing one over the other.
Does the laser damage the sample?
Generally, no. We use very low power levels for sensitive materials. However, if you use a high-powered laser on a dark, light-absorbing sample, it can heat up or even burn. A skilled operator knows how to balance the laser power to get a good signal without damaging the integrity of the material being tested.
Can the Raman Method identify every substance?
While it is incredibly versatile, it isn’t perfect. Pure metals, for example, do not have the molecular vibrations required for a Raman signal. Also, as mentioned before, highly fluorescent materials can sometimes hide their signal. However, for the vast majority of organic and inorganic compounds, it is extremely effective.
How expensive is it to implement the Raman Method?
The cost has dropped significantly over the last decade. While high-end research-grade systems still cost six figures, portable and handheld units are now affordable for many small businesses and local law enforcement agencies. The return on investment is usually very high because of the savings in time and sample preparation costs.
The Raman Method is essentially the ultimate translator for the molecular world. It takes the chaotic vibrations of atoms and turns them into a clear, readable map that tells us exactly what we are looking at. From the deep reaches of space to the medicine in your cabinet, this technique is quietly providing the data that keeps our modern world moving forward. It is a perfect blend of physics, chemistry, and practical engineering that continues to surprise me even after ten years in the field.