
Can FTIR detect hydrogen bond dissociation? | ResearchGate
Invisible Molecules: Critical Detection Failures in FTIR Spectroscopy
I’ve spent over a decade squinting at interferograms and arguing with stubborn benchtop units, and if there’s one thing I’ve learned, it’s that FTIR spectroscopy is a bit of a diva. It’s incredibly powerful for identifying organic compounds, sure, but it has these massive, glaring blind spots that catch junior analysts off guard every single time. Look—just because the machine didn’t beep doesn’t mean your sample is pure.
Understanding What Can Ftir Not Detect is actually more important than knowing what it can. If you walk into a lab thinking this technique is a universal “tell-me-everything” tool, you’re going to report results that are, frankly, embarrassing. It’s a molecular vibration tool, not a magic wand. If the physics of the bond don’t align with the physics of the light, you’re basically staring into a black hole.
Honestly? I love this technique. But its limitations are hard-coded into the laws of thermodynamics and quantum mechanics. You can’t “software update” your way out of a missing dipole moment.
Let’s get into the weeds of why this “gold standard” tool leaves so much on the table. It’s a big deal if you’re trying to solve a real-world contamination problem or verify a chemical synthesis. Sometimes, the most important part of your sample is exactly what the detector refuses to see.
The Symmetry Problem and Dipole Moment Blindness
The most fundamental rule of FTIR spectroscopy is that a molecular vibration must cause a change in the net dipole moment to be IR active. If the vibration is perfectly symmetrical, the infrared light just passes right through like it isn’t even there. It’s invisible. Seriously, you could have a jar full of nitrogen gas and the FTIR would tell you it’s looking at a vacuum.
This is where What Can Ftir Not Detect starts: homonuclear diatomic molecules. We’re talking about the heavy hitters like Oxygen (O2), Nitrogen (N2), and Hydrogen (H2). Because these molecules consist of two identical atoms, their stretching vibration doesn’t change the charge distribution. No change in dipole means no absorption peak. If your process gas is leaking nitrogen into your sample chamber, your FTIR is blissfully unaware.
Homonuclear Diatomics and Atmospheric Silence
In a practical lab setting, this means you can’t use FTIR to measure the purity of your welding gas or the oxygen content in a sealed vial. These gases are “IR silent.” It’s a funny quirk of nature that the two gases making up 99 percent of our atmosphere don’t interfere with the spectrum, yet carbon dioxide—a tiny fraction of the air—screams like a banshee at 2350 cm-1. It’s a blessing for atmospheric compensation, but a curse for elemental gas analysis.

(PDF) Materials Today May 2015 Identify Defects and Characterize With …
I’ve seen people try to quantify hydrogen gas levels using a high-end spectrometer, only to realize they were chasing ghosts. Unless you’re using Raman spectroscopy, which relies on polarizability rather than dipole moments, those symmetrical bonds are going to stay hidden. It’s the ultimate “hide and seek” champion of the molecular world.
Symmetry in Complex Organic Structures
It isn’t just simple gases, either. If you have a highly symmetrical organic molecule, like certain trans-alkenes or specific para-substituted benzenes, some of those key vibrations will be “IR forbidden.” The molecule is vibrating, but because the movement is perfectly balanced on both sides, the dipole moment stays at zero. You end up with a spectrum that looks much simpler than the molecule actually is.
This “missing information” is why we often pair FTIR with Raman. They are complementary; what one misses, the other usually catches. But if you only have an FTIR bench, you have to be very careful about assuming a clean spectrum equals a simple molecule. It might just be a very symmetrical one.
Elemental Analysis and Inorganic Hurdles
Another massive hurdle in What Can Ftir Not Detect is the realm of pure elements and simple inorganic salts. FTIR is a molecular bond tool. It does not “see” atoms. If you want to know how much lead is in your paint or how much arsenic is in your water, FTIR is the wrong tool for the job. It won’t tell you that there’s Gold (Au) or Silver (Ag) in a sample because those are atomic structures, not molecular ones with IR-active vibrations.
Even when you get into compounds, many common inorganic salts are invisible in the standard mid-IR range. Think about table salt (NaCl) or Potassium Bromide (KBr). These materials are so “invisible” to IR light that we actually use them to make the windows and pellets that hold the samples! If the spectrometer could see them, we wouldn’t be able to see through them to the sample.
Single Atom Detection Failures
Look—if it doesn’t have a covalent bond that vibrates between 400 and 4000 cm-1, the FTIR is basically blind. Noble gases like Argon or Helium? Forget about it. Metals in their metallic state? They just reflect the light like a mirror. This is why metallurgical labs use ICP-OES or XRF instead of infrared spectroscopy. They need to see the elements; we’re just here for the functional groups.
I once had a client ask me to find “mercury contamination” in a polymer using FTIR. I had to explain that unless that mercury is bonded to a carbon or an oxygen in a very specific way, the FTIR won’t show a single peak for it. You could have a bead of liquid mercury sitting in the sample tray and the spectrum would just show the background air.
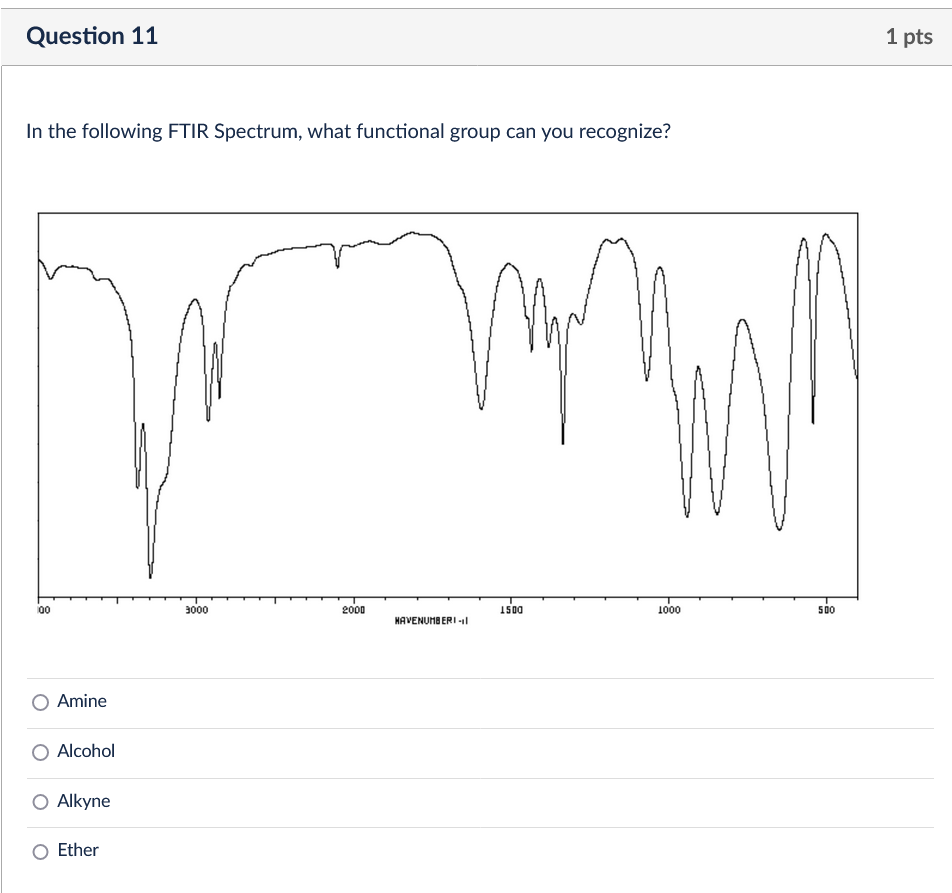
Solved In the following FTIR Spectrum, what functional group | Chegg.com
The Problem with Ionic Bonds
Ionic bonds don’t behave like the “springs” we talk about in organic chemistry. In a crystal lattice of something like Sodium Chloride, the vibrations happen at much lower frequencies than a standard FTIR can reach. Most commercial units cut off at 400 cm-1. The “lattice vibrations” of many minerals and salts happen in the far-infrared or “terahertz” region. If you’re using a standard lab setup, these materials are effectively transparent.
This is why you can’t easily distinguish between different types of sand or simple salts without specialized optics. The FTIR is designed for the world of Carbon, Hydrogen, Oxygen, and Nitrogen. Once you move into the bottom half of the periodic table, the tool becomes significantly less useful. It’s just not what it was built for.
Concentration Limits and Matrix Interference
We need to talk about the “LOD” or Limit of Detection. People think FTIR can find a needle in a haystack. It can’t. Honestly, it’s actually pretty bad at trace analysis. If you have a contaminant that makes up less than 0.1% of your sample, What Can Ftir Not Detect becomes a very long list. The main signals from the “bulk” material will simply drown out the tiny signals from the contaminant.
This is exacerbated by “matrix interference.” If you’re looking for a small amount of oil in water, the water signal is so massive and broad that it physically overlaps and hides the oil peaks. Water is the “bully” of the IR spectrum. It absorbs so strongly that it can opaque the entire detector if the path length is more than a few microns.
Trace Analysis Constraints
In my experience, FTIR is a bulk identification tool. If you’re looking for parts-per-million (ppm) or parts-per-billion (ppb) levels of a substance, you need to stop. You’re in GC-MS or HPLC territory now. Trying to find 50 ppm of a plasticizer in a PVC sample using FTIR is like trying to hear a whisper at a heavy metal concert. It’s technically there, but the “noise” of the main signal makes it impossible to resolve.
There are techniques to help, like Attenuated Total Reflection (ATR) or solvent extraction, but even then, you’re pushing the limits. The dynamic range of FTIR is nowhere near as broad as chromatographic methods. It’s great for “what is this chunk of plastic?” but terrible for “is there a trace of poison in this water?”
Water Vapor and Carbon Dioxide Distortions

How To Use Ftir Machine at Hillary Mccarty blog
Even the air inside the spectrometer can prevent you from detecting what you need. If you don’t purge the system with dry nitrogen, the water vapor and CO2 in the air will create “noise” in the very regions where many important bonds appear (like O-H stretches or C=O bonds). If your sample has a weak peak in the same spot as a CO2 “spike,” you’re never going to see it.
- Water Vapor: Masks the 3600-3900 cm-1 and 1500-1800 cm-1 regions.
- Carbon Dioxide: Creates massive interference at 2350 cm-1 and 667 cm-1.
- Path Length Issues: Liquid samples thicker than 25 microns often “bottom out” the signal.
- Secondary Peaks: Overtones and combination bands can create “ghost peaks” that look like contaminants but aren’t.
If you aren’t careful with your background subtraction, the FTIR will “detect” the humidity of the room rather than the chemistry of your sample. It’s a common rookie mistake that leads to very confusing reports.
Physical State and Optical Challenges
Finally, there’s the physical reality of the sample. FTIR relies on light passing through or reflecting off a surface. If the light can’t get back to the detector, you get nothing. This brings us to a huge category of What Can Ftir Not Detect: non-reflective, highly scattering, or totally opaque materials. If you have a sample that is “carbon black” filled (like a car tire), the carbon absorbs almost all the IR radiation across the entire spectrum. The result? A flat line.

Infrared spectroscopy(IR) & FTIR (Analytical Technique) | PPTX
The same goes for very rough surfaces. If the sample scatters the light in every direction except toward the detector, the signal-to-noise ratio drops to zero. You might have the most IR-active compound in the world, but if it’s trapped in a scattering matrix, it remains invisible to the spectrometer.
Reflective Surface Interference
Paradoxically, being too reflective is also a problem. If you try to analyze a thin coating on a shiny metal surface using standard transmission, the light just bounces off the metal and never goes through the coating. You need specific techniques like Reflection-Absorption (RAIRS) to see these. Without the right accessory, those coatings are effectively invisible.
Look—the geometry of the sample matters just as much as the chemistry. If you just drop a piece of uneven rubble onto an ATR crystal and don’t apply enough pressure, you’ll get a weak, useless spectrum. The FTIR isn’t failing because the molecules aren’t there; it’s failing because the optical contact is poor.
Particle Size and Scattering
If you’re working with powders, the particle size can ruin your day. If the particles are roughly the same size as the wavelength of the IR light (about 2 to 15 microns), you get a phenomenon called the Christiansen Effect. This causes massive distortions in the peak shapes and can even make peaks disappear or invert. It’s an optical artifact that hides the true molecular signature.
-
- Opaque Solids: Materials like coal or heavily dyed plastics absorb too much light.
- Scattering Powders: Large grains deflect light away from the detector.
- Aqueous Solutions: The water signal overwhelms almost everything else.

(Color online) Example of the experimental FTIR transmittance and …
- Gas Phase Mixtures: Low-pressure gases often have concentrations too low for standard detectors.
In all these cases, the chemistry is there, but the FTIR is effectively blind due to the physical state of the sample. You have to prep the sample perfectly, or you’re just looking at noise.
Common Questions About What Can Ftir Not Detect
Can FTIR detect pure metals like Gold or Platinum?
No, FTIR cannot detect pure metals. Metals do not have molecular bonds that vibrate in the infrared range; instead, they have “free electrons” that reflect infrared radiation. If you put a piece of gold in an FTIR, it will act like a mirror and reflect the beam, providing no spectral information about the metal itself.
Does FTIR work for identifying nitrogen or oxygen gas levels?
Standard FTIR cannot detect N2 or O2 because they are homonuclear diatomic molecules. These molecules do not undergo a change in dipole moment when they vibrate, making them “IR inactive.” To detect these gases, you would typically use Raman spectroscopy or Gas Chromatography.
Why is it hard to see trace contaminants with FTIR?
FTIR spectroscopy generally has a detection limit around 0.1% to 1% by weight. Because it is a bulk analysis tool, the signal from the main component (the matrix) usually overwhelms the tiny signal from a trace contaminant. If the contaminant is present at parts-per-million levels, it is usually “invisible” to the spectrometer.
Can FTIR distinguish between different isotopes of the same element?
Actually, FTIR can sometimes see isotope shifts, but it’s very difficult. Replacing Hydrogen with Deuterium, for example, changes the mass of the “spring” in the molecular vibration, which shifts the peak significantly. However, for heavier elements, the shift is so small that a standard lab spectrometer usually cannot resolve the difference.
Understanding these boundaries is what separates a technician from an expert. FTIR is a powerful ally, but only if you know when to stop relying on it and reach for a different tool in the shed.